My latest publication shows that over three-quarters of people having chemotherapy in New South Wales experience multiple side effects during their treatment, and for over 60% of people this included a serious side effect. These results confirm previous research that suggests side effects might be more common, and more serious, in clinical practice (ie ‘real life’) than reported in clinical trials.
During their chemotherapy, 86% of our sample (who had lung, breast or colorectal cancer) reported at least one side effect, and 67% experienced six or more different side effects. Fatigue was the most common side effect (80%), followed by pain (75%), constipation (74%), and diarrhoea (74%). For nearly a quarter of participants (24%) the side effects were mild, but for many more (62%) the side effects were moderate or severe.
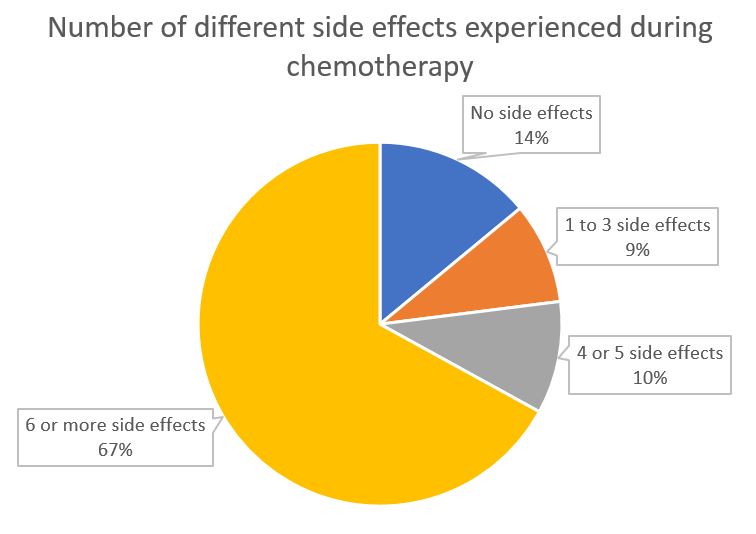
The number of different self-reported side effects experienced during chemotherapy by participants with lung, breast or colorectal cancer in the EOCC study
Older people in our sample were less likely to have a side effect. This is perhaps because older people tend to receive less aggressive treatments, despite this also possibly reducing how effective their chemotherapy is. Other things, like the type and stage of cancer, gender, education, and socioeconomic status did not change how likely a person was to have a side effect.
When we looked at the patterns of side effects over time, many people had mild side effects which stayed with them throughout their chemotherapy, especially constipation, diarrhoea, mucositis and nausea / vomiting. There was also a particularly large proportion of people reporting serious fatigue throughout their treatment.
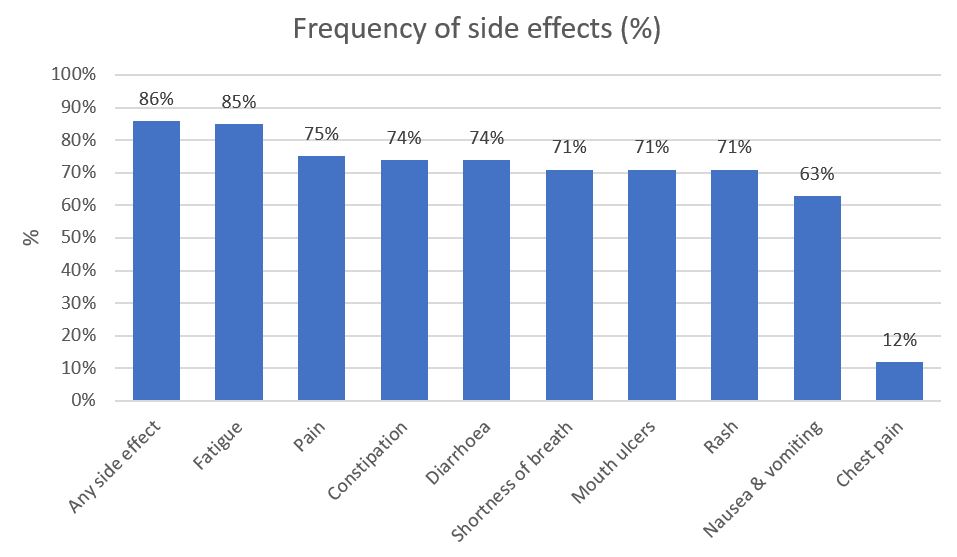
The frequency of side effects self-reported side effects experienced during chemotherapy by participants with lung, breast or colorectal cancer in the EOCC study
The first study of this type in Australia, our Elements of Cancer Care study followed 441 people with breast, lung and colorectal cancer having chemotherapy in New South Wales. We interviewed them each month during their chemotherapy treatment to ask them about a wide range of topics, including what side effects they’d experienced and how serious they were. We also collected information from their medical records at the hospital and with Medicare.
Side effects in real life vs clinical trials
When new chemotherapy treatments are developed, the side effects they cause are tested in research studies called clinical trials. Doctors, patients and policy makers then base their decisions about chemotherapy on the data from these clinical trials.
But, what happens in clinical trials does not always reflect what happens in real life. Clinical trials usually have very strict criteria for who can participate. Clinical trial participants are usually younger and fitter than typical cancer patients, and so be more able to cope physically with chemotherapy and therefore less likely to have a side effect. In addition, clinical trials are usually conducted in large, high-quality, teaching hospitals with extra monitoring and treatment of side effects, which may reduce how often they occur, or how serious they become.
Asking patients about side effects
How patients are asked about side effects can also influence what they report. When doctors or nurses ask general questions like “how have you been feeling” or “have you had any side effects” patients might not remember or report all of their side effects, particularly if they are not still happening. We gave participants a checklist of side effects, which may have encouraged them to report a greater variety of side effects, and those side effects which were less severe. This is a technique which could be implemented by doctors and nurses in cancer care clinics.
What does this mean?
On top of dealing with a diagnosis of cancer and being treated with chemotherapy, having side effects can affect someone’s physical health, survival, quality of life and emotional state. Because our information comes from real life, rather than clinical trials, it allows doctors, nurses, policy makers and patients to think more realistically about the side effects of chemotherapy.
Full reference (Open Access): Pearce A, Haas M, Viney R, Pearson SA, Haywood P, Brown C, Ward R (2017). Incidence and severity of self-reported chemotherapy side effects in routine care: A prospective cohort study. PLOS One 12(10): e0184360
The Elements of Cancer Care study was funded by the National Health and Medical Research Council (Health Services Research Grant ID 455366). Alison was supported by a University of Technology Sydney Doctoral Scholarship, and a PhD top-up scholarship from NHMRC Health Services Research Grant (ID455366). Sallie is supported by a Cancer Institute NSW Career Development Fellowship (ID: 12/CDF/2-25). No funding organisation had any role in the study design, data collection and analysis, decision to publish or preparation of the manuscript.